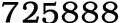|
1.Ammonium acetate Chemical Properties Name:Ammonium acetate mp: 110-112 °C (dec.)(lit.) density :1.07 g/mL at 20 °C Fp:136 °C storage temp:2-8°C solubility H2O:1 M at 20 °C, clear, colorless Water Solubility :1480 g/L (20 ºC) Merck:14,495 BRN:4186741 CAS Registry number:631-61-8 NIST Chemistry Reference:Ammonium acetate, anhydrous(631-61-8) EPA Substance Registry System:Acetic acid, ammonium salt(631-61-8) 2.Ammonium acetate Usage And Synthesis Chemical Properties: white adhering crystals General Description: A white crystalline solid. The primary hazard is the threat to the environment. Immediate steps should be taken to limit its spread to the environment. Ammonium acetate is used in chemical analysis, in pharmaceuticals, in preserving foods, and for other uses. Air & Water Reactions: Water soluble. Reactivity Profile: Ammonium acetate causes the decomposition of sodium hypochlorite within a few seconds. Health Hazard: Inhalation of dust irritates nose and mouth. Ingestion irritates mouth and stomach. Contact with dust causes irritation of eyes and mild irritation of skin. Fire Hazard: Special Hazards of Combustion Products: Irritating vapors of ammonia acetic acid, and nitrogen oxides may form in fires. 3.Safety Information Safety Statements:24/25 RIDADR UN:9079 WGK Germany:3 RTECS: AF3675000 F:3 HS Code:29152900 Hazardous Substances Data:631-61-8(Hazardous Substances Data)
Related Articles -
Ammonium, acetate,
|