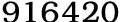|
Orlistat is used in the treatment of obesity and was approved by the European Commission in 1998. It is a selective inhibitor of gastric and pancreatic lipase and leads to a reduction in dietary fat lipolysis and absorption.1 Although mild but unpleasant gastrointestinal side effects are commonly reported with orlistat use there is also concern that it may be associated with an increased risk of serious hepatic events. The US Food and Drug Administration first issued a warning about a possible link between orlistat(CAS NO:96829-58-2) and serious liver injury in 20092 3 based on an analysis of 32 case reports received between 1999 and 2008. The European Medicines Agency has also reviewed the strength of evidence relating to this possible risk.4 The analysis of individual case reports is often unable to give reliable conclusions about causality and a range of population based studies are needed to help inform decisions about the likely risks and benefits of this chemical. A meta-analysis of clinical trial data involving around 10000 patients found no evidence that orlistat was associated with increases in selected variables of liver function,5 and a likely mechanism of action has not been identified. Assessments by both agencies concluded that there was no strong evidence to determine a causal association. However, results from clinical trials do not always reflect the effects of drugs when used in the general population, and to date no studies have been published based on orlistat use in the real world. It remains the only drug treatment option for obesity, is widely used, and in many countries is available without prescription. Further clarity on safety is therefore needed. People using orlistat are likely to have a different underlying risk of acute liver events from non-users owing to obesity and its comorbidities, and many study designs would be unable to fully account for these important differences. We conducted a self controlled case series study in which each participant acts as his or her own control to avoid the inherent biases caused by differences between people. Since early 2000 reports of liver injury associated with orlistat have accumulated, raising concerns about its safety. Although randomised trials found no such association, observational studies based on real world orlistat use are lacking.In a large population based cohort, the rate of adverse liver events seems to be temporarily increased both immediately before and immediately after starting treatment with this chemical. This suggests that the increased risk of liver injury linked to orlistat may reflect changes in underlying health status associated with the decision to start treatment, rather than a causal effect of the drug. Obesity has become a major health problem worldwide and orlistat is currently the only available drug intervention of proved efficacy. Possible safety concerns about orlistat are therefore of great importance. By using the self controlled case series design we were able to establish that the risk of liver injury was increased to a similar degree both just before and just after starting treatment, strongly suggesting that the association is non-causal. Our results imply that studies based only on traditional epidemiological designs or spontaneous adverse events are likely to detect associations between starting orlistat treatment and liver injury, but such associations should not be interpreted as evidence for an adverse causal effect of the drug unless an increased risk in the period immediately before the start of treatment can be excluded. Want to learn more information about Orlistat, you can access the guidechem.com. Guidechem.com is just a place for you to look for some chemicals.
Related Articles -
Orlistat, guidechem, 96829-58-2,
|