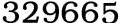|
chemistry answers Understanding closure property is always challenging for me but thanks to all math help websites to help me out closure property">absolute deviation in chemistry Introduction to absolute deviation in chemistry: According to mathematical expression the term absolute deviation is defined as an aspect of informational data which determines the difference between the element and the given point. In general, the position from which the divergence is calculated. Referred to as the rate of either mean or median of the data set. Absolute Deviation in Chemistry- Determination of Relative Standard Deviation According to statistical data or probability theory, the relative standard deviation (RSD or percentage of RSD) is determined as the complete value of the coefficient of variation. It is frequently spoken as a percentage. Sometimes the similar term used is the relative variance which is the square of the coefficient of variation. As well, as the relative standard error is a measure of a statistical estimate’s consistency obtained by separating the standard error by the estimation; then multiplies by 100 to be articulated as a percentage. The relation for the standard deviation is extensively used in analytical chemistry to convey the exactness and repeatability of an assay. The mean standard deviation of an assay is determined as X, which is then multiplied by 100. Is then divided by average of an array X gives the relative standard deviation. Is given in terms of an equation: (standard deviation of array X) × 100 / (average of array X) = relative standard deviation. General terms for absolute deviation in chemistry: Certain other factors include deviation in chemistry such as accuracy, absolute temperature , absolute error, absolute zero, accuracy, Celsius, deviation, base unit, Centi, amperes, calibration, etc. The absolute deviation in chemistry as follows Absolute error: Absolute improbability relies with absolute error. Absolute error is generally used to explain inaccuracies. Note down that when fixed errors are related with undetermined errors. Absolute temperature: The absolute temperature is usually measured in terms of scalar units. That is determined as absolute zero. In SI system the absolute temperature is measured in terms of the Kelvin scale. Absolute zero: It is basically measured when the amount of an ideal gas becomes zero. The absolute zero is defined as the zero according to the Kelvin scale, i.e. -273.15°C on the Celsius scale, and then -459.67°F on the Fahrenheit scale. Accuracy: It is determined as the correctness of a single measurement. These are some of the individual measurements for the absolute deviations in chemistry. like to share with you all through my article chemistry answers Understanding closure property is always challenging for me but thanks to all math help websites to help me out closure property
Related Articles -
Absolute, Deviation, in, Chemistry,
|