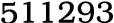|
Zinc chloride is the name of chemical compounds with the formula ZnCl2 and its hydrates. Zinc chlorides, of which nine crystalline forms are known, are colorless or white, and are highly soluble in water. ZnCl2 itself is hygroscopic and even deliquescent. Samples should therefore be protected from sources of moisture, including the water vapor present in ambient air. Zinc chloride finds wide application in textile processing, metallurgical fluxes, and chemical synthesis. No mineral with this chemical composition is known aside from a very rare mineral, simonkolleite, Zn5(OH)8Cl2·H2O. 1. Behavior and Chemical Properties Very soluble in water, also in ethanol and acetone. With excess water, acidic (pH ~ 4) zinc oxychloride is formed. Zinc chloride has a rather low melting point and sublimed easily. This indicates that it has strong molecular character rather than ionic. Electron diffraction studies show that zinc chloride has linear Cl-Zn-Cl structure in the gas phase . Concentrated , aqueous solution dissolves starch and cellulose and silk. Hence the solution attacks paper and cannot be filtered through a filter paper. 2. History and Uses The antiseptic nature of Zinc chloride has found its use in antiseptic, astringent and in treating ulcers and pododermatitis. It is also used in preserving anatomical specimens, wood preservatives, deodorant, disinfecting and embalming materials. The chloride is used as fluxes on copper alloys and, with hydrochloric acid added, for stainless steels. It is also used as a mordant in printing and dyeing textiles and in the vulcanizing processes of fiber and rubber. It is used as an electrolyte in dry cell batteries, and in metal industry, in galvanizing iron and as an electrolyte for electroplating. In chemical synthesis, the anhydrous zinc chloride is used as a dehydrating agent. And as a standard saturated solution, it is used for calibration of the electrohygrometer in oil industry. 3. Hazard, Storage and Handling Potential symptons of over exposure to fumes are conjunctivitis, irritation of eyes, skins and throat. May also cause chest pain, pulmonary fibrosis and skin burns. Chronic effects may include stomach pains, vomiting, diarrhea, chest pain, edema and possibly mutagenic effects. The oral lethal dose 50 percent kill for rats (ORL-RAT LD50) is about 350 mg/kg. Zinc chloride is very hygroscopic and the anhydrous must be kept in air-tight container, in a cool dry place. Ensure good ventilation (such as in fume cupboard) when using the chemical, wearing rubber gloves and safety goggles. If ingested, take large quantities of milk or water and seek medical advice. In case of contact with skins apply baking soda to neutralize the acidic effects. When contact with eyes, splash with plenty of water and seek medical attention.
Related Articles -
Zinc chloride, 7646-85-7,
|