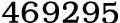|
Sulfamic acid, also known as amidosulfonic acid, amidosulfuric acid, aminosulfonic acid, and sulfamidic acid, is a molecular compound with the formula H3NSO3. This colorless, water-soluble compound finds many applications. Sulfamic acid melts at 205 °C before decomposing at higher temperatures to H2O, SO3, SO2, and N2. Sulfamic acid is a dry, non-volatile, non-hygroscopic, odorless, white crystalline, stable acid .It is moderately soluble in water and forms strongly acidic aqueous solution that is comparable in acidity to the common strong mineral acids but it may be easily handled and stored in the dry form. At room temperature ,dilute aqueous sulfamic acid solution is stable for a long time but rapid hydrolysis occurs at elevated temperatures .Its solution is less corrosive toward metals than other mineral acid. Because of advantages such as handling ease, solubility and low corrosiveness, sulfamic acid is used for many diversified applications. Name:Sulfamic acid EINECS:226-218-8 Molecular Formula:H3NO3S CAS Registry Number:5329-14-6 Synonyms:sulphamic acid; Amidosulfonic acid; Amidosulfuric acid; Aminosulfonic acid InChI:InChI=1/H3NO3S/c1-5(2,3)4/h(H3,1,2,3,4) HS Code:28111980 Appearance:white crystalline solid Molecular Weight:97.09 Density:2.126 Boiling Point:247 Melting Point:215-225? (dec.) Flash Point:205 Storage Temperature:Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances. Refractive index:1.553 Solubility:146.8 g/L (20 °C) in water Stability:Stable. Usage:As standard in alkalimetry, in acid cleaning, in nitrite removal, in chlorine stabilization for use in swimming pools, cooling towers, paper mills. Chemical Properties: white crystals General Description: A white crystalline solid. Density 2.1 g / cm3. Melting point 205°C. Combustible. Irritates skin, eyes, and mucous membranes. Low toxicity. Used to make dyes and other chemicals. Air & Water Reactions: Moderately soluble in water . Reactivity Profile: Sulfamic acid reacts exothermically with bases. Aqueous solutions are acidic and corrosive. Health Hazard: TOXIC; inhalation, ingestion or skin contact with material may cause severe injury or death. Contact with molten substance may cause severe burns to skin and eyes. Avoid any skin contact. Effects of contact or inhalation may be delayed. Fire may produce irritating, corrosive and/or toxic gases. Runoff from fire control or dilution water may be corrosive and/or toxic and cause pollution. Fire Hazard: Non-combustible, substance itself does not burn but may decompose upon heating to produce corrosive and/or toxic fumes. Some are oxidizers and may ignite combustibles (wood, paper, oil, clothing, etc.). Contact with metals may evolve flammable hydrogen gas. Containers may explode when heated.
Related Articles -
Sulfamic acid, 5329-14-6,
|